|
|
Recombinant Histone H3.3-H4 tetramers
Catalog # : EPX-04-RH
Source : Human
Expressed in : E. Coli
Quantity : 50ug of recombinant H3.3-H4 tetramers at 1ug /ul
Background:
Histone H3.3 associates with H4 to form an H3.3-H4 tetramer, a core component of the nucleosome (1). The nucleosome is the basic unit of chromatin and consists of 147 base pairs of DNA wrapped around an octamer of core histones H2A, H2B, H3 and H4 (2). The histone variant H3.3 replaces conventional H3.1 in a wide range of nucleosomes in active genes and constitutes the predominant form of histone H3 in non-dividing cells and is incorporated into chromatin independently of DNA synthesis (1, 3).
Protein details:
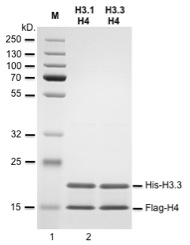
Recombinant human histone H3.3-H4 dimers were produced in E. Coli as fusion proteins with an N-terminal 6xHis or FLAG epitopes, purified using FPLC and formulated in a storage buffer containing 20mM Tris-Cl pH 7.9, 1mM EDTA, 1M NaCl, 0.5mM PMSF and 1mM DTT. Protein concentration was determined by spectrometry.
Quality control:
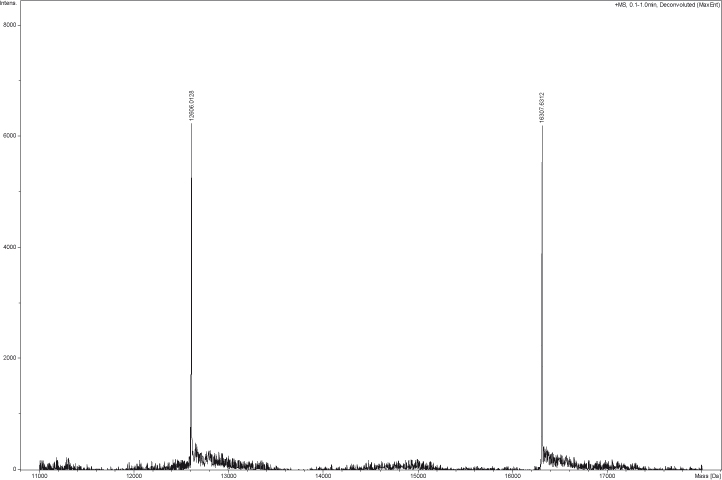
Each lot has been evaluated by ESI-TOF analysis and 12% SDS-PAGE (NuPAGE MOPS SDS, Invitrogen).
Application Notes:
For research use only.
Storage:
-80°C
Guarantee:
Products guaranteed stable for 2 years from date of receipt when stored properly.
Purity:
>98% purity by SDS PAGE.
Protein sequences: 6xHis-H3.3 (Theoretical Mw: 16439.02) MGSHHHHHHGSARTKQTARKSTGGKAPRKQLATKAARKSAPSTGGVKKPH RYRPGTVALREIRRYQKSTELLIRKLPFQRLVREIAQDFKTDLRFQSAAI GALQEASEAYLVGLFEDTNLCAIHAKRVTIMPKDIQLARRIRGERA
Flag-H4 (Theoretical Mw: 12717.75) MIDYKDDDDKLESGRGKGGKGLGKGGAKRHRKVLRDNIQGITKPAIRRLA RRGGVKRISGLIYEETRGVLKVFLENVIRDAVTYTEHAKRKTVTAMDVVY ALKRQGRTLYGFGG
References: 1. Drané et al., (2010). Genes Dev. Jun 15;24(12):1253-65. 2. Luger et al., (1997) Nature, 389(6648):251-60. 3. Ahmad and Henikoff. (2002). Mol Cell. Jun;9(6):1191-200. |
|---|
© 2011-2016 EpiGex. All rights reserved